hCFB
Nomenclature
C57BL/6JSmo-Cfbtm1(hCFB)Smoc
Cat. NO.
NM-HU-2000078
Strain State
Repository Live
Gene Summary
Gene Symbol
Cfb
Model Description
Validation Data

Fig1. Detection of CFB expression in liver by RT-PCR.
Wild type: only one band at 441 bp with primers F1/R1(mCfb);
Homozygous: only one band at 442 bp with primers F2/R2(hCFB);
Abbr. M, DNA marker; HO, homozygous; WT, wild type.

Fig2. Detection of human and mouse CFB expression in serum by ELISA (n=2).
Abbr. HO, homozygous; WT, wild type.
Note: The mouse CFB ELISA kit cross-reacted with mCFB and humanized CFB.
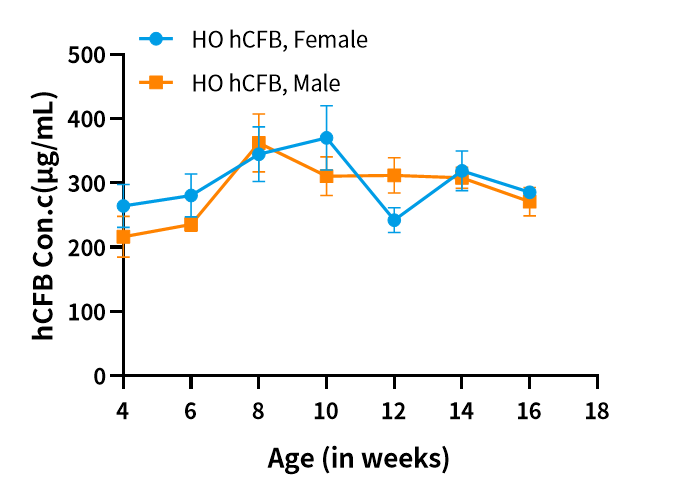
Fig3. Detection of hCFB expression in serum by ELISA(n=3/group).
Abbr. HO, homozygous; WT, wild type

Fig4. Body Weight(A) and Survival Curve(B) of WT C57BL/6 and HO hCFB mice.
Abbr. HO, homozygous; WT, wild type.

Fig5. Blood Biochemistry of WT C57BL/6 and HO hCFB mice.
Abbr. HO, homozygous; WT, wild type.

Fig6. Detection of liver CFB mRNA remaining at Day 7 (A) and serum protein level (B) after single dose of test drug (n=3/group). (Data from a collaborator)

Fig7.Partial representative pictures of liver and kidney pathology in hCFB knockin mice. No obvious lesions were observed in hCFB knockin mice.
You may also like
Shanghai Model Organisms Center Inc has licensed CRISPR-Cas9 technology from Broad Institute
On Dec 16, 2018, Broad Institute and Shanghai Model Organisms Center Inc (SMOC) has entered into a non-exclusive license agreement under which Broad has granted SMOC worldwide rights to commercialize a service platform for genetically modified mouse models under Broad's intellectual property.
Learn moreWorkshop:Progress and Advances in Preclinical immuno-Oncology Research
SMOC’s Annual Progress and Advances in Preclinical immuno-Oncology Research: The workshop is designed as a forum for ideas and opinions exchange on how to decrease the rate of clinical failures in oncology and immuno-oncology.
Learn moreCustomizing Mouse Models: Delivering in 100 Days to Speed Up Your Research!
At GenoBioTX, we understand that the lengthy wait times for gene-modified mouse models can hinder your research progress. Traditional methods often require 6-9 months, leading to delays and increased costs. That’s why we’re thrilled to introduce our innovative service designed to streamline this process and deliver results faster.
Learn more


